 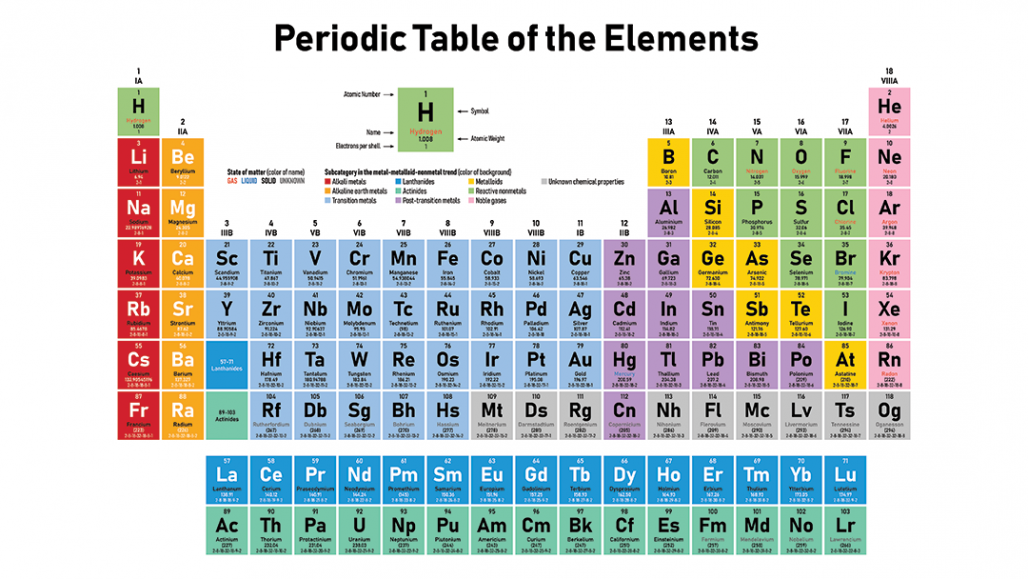
Each element is identified by the number of protons in its atoms. It is typically located as a subscript to the left of the element symbol. There are 118 elements on the periodic table. The atomic number is the smaller of the two numbers in the symbol. 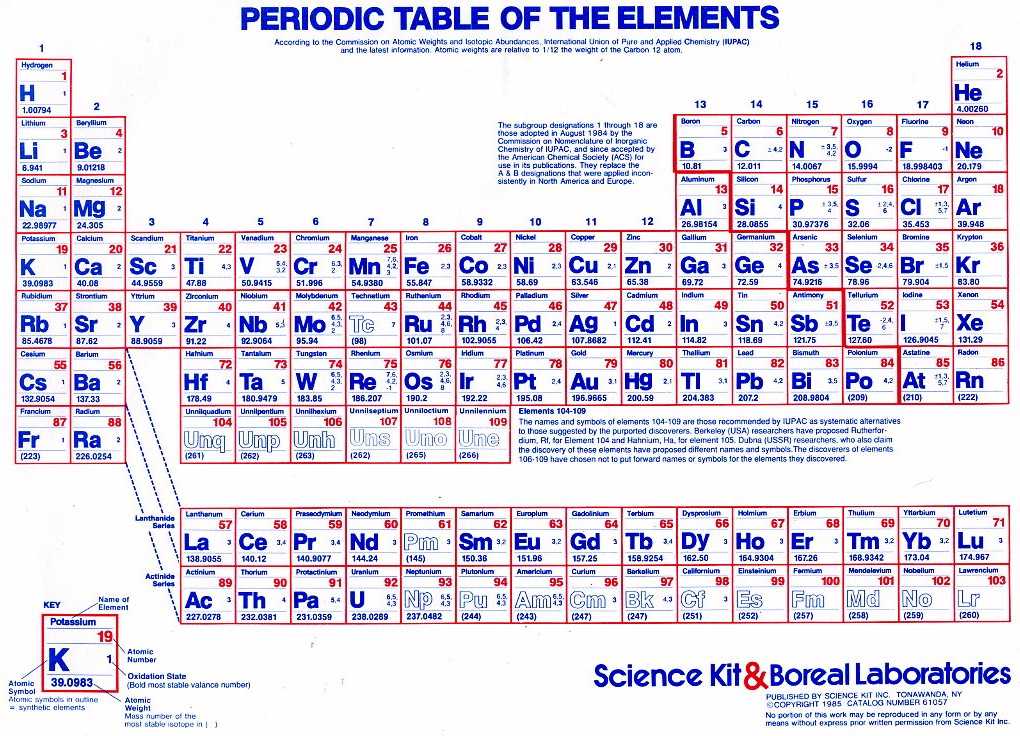
For example, if the symbol is written as 14 6C, the number "6" is listed. More commonly, the isotope symbol already tells you the atomic number.The atomic weight or mass shows the average size of an atom. The periodic table is arranged numerically according to atomic number. It tells you how many protons the element has. Please note that the elements do not show their natural relation towards each other as in the Periodic system. The first chemical element is Hydrogen and the last is Ununoctium. For example, if the symbol is 14C, you know the element symbol is C or that the element is carbon. The atomic number is the number above the symbol. For chemistry students and teachers: The tabular chart on the right is arranged by Atomic number. What is atomic weight The nuclei of atoms also contain. All of these elements display several other trends and we can use the periodic law and table formation to predict their chemical, physical, and atomic properties. Interactive periodic table with up-to-date element property data collected from authoritative sources. You can use the symbol to look up the number. Thus, all atoms that have the same number of protons-the atomic number-are atoms of the same element. The elements in the periodic table are arranged in order of increasing atomic number. There is more than one way to write an isotope symbol, but the element symbol will always be included. You can find the atomic number from an isotope symbol.For example, if you are told the element name is aluminum, you can find the name or symbol Al to determine the atomic number is 13. The table was named the periodic table because similar. The periodic table is a tabular array of the chemical elements organized by atomic number, from the element with the lowest atomic number, hydrogen, to the. In calculations, it is denoted by the capital letter Z. All the elements of the periodic table are arranged in order of increasing atomic (proton) number. For this reason, it's sometimes called the proton number. For a single atom of an element, this would be a whole number, adding the number of protons, neutrons, and electrons together for the atom. The atomic number is simply the number of protons in an atom. Most periodic tables include a value for atomic mass (also called atomic weight) on each element tile. In fact, this number is how you can distinguish one element from another. While other numbers may be decimal values, the atomic number is always a simple positive whole number. Each element on the periodic table has its own atomic number. There may be many numbers on a periodic table, so how do you know which one to pick? The atomic numbers go in order on the table. If you have an element name or symbol, use a periodic table to find the atomic number.When an organism dies, it stops taking in carbon-14, so the ratio of carbon-14 to carbon-12 in its remains, such as fossilized bones, will decline as carbon-14 decays gradually to nitrogen-14 2 ^2 2 squared. The elements are arranged on the table in order of increasing number of protons in the nucleus. As animals eat the plants, or eat other animals that ate plants, the concentrations of carbon-14 in their bodies will also match the atmospheric concentration. atomic number, the number of a chemical element in the periodic system and on the periodic table that equals the number of protons in the nucleus of the atom. A chemical element, often simply called an element, is a type of atom which has the same number of protons in its atomic nucleus (i.e., the same atomic number, or Z). As plants pull carbon dioxide from the air to make sugars, the relative amount of carbon-14 in their tissues will be equal to the concentration of carbon-14 in the atmosphere. This is a list of the 118 chemical elements that have been identified as of 2023. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. These forms of carbon are found in the atmosphere in relatively constant proportions, with carbon-12 as the major form at about 99%, carbon-13 as a minor form at about 1%, and carbon-14 present only in tiny amounts 1 ^1 1 start superscript, 1, end superscript. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.The chemical symbol for Hydrogen is H. For example, carbon is normally present in the atmosphere in the form of gases like carbon dioxide, and it exists in three isotopic forms: carbon-12 and carbon-13, which are stable, and carbon-14, which is radioactive. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
